Real World Research
At Optimapharm, we recognise that generating evidence beyond the controlled environment of clinical trials is critical for product success. Our real-world research services are designed to bridge clinical development and commercialisation, ensuring our clients can demonstrate value to regulators, payers, healthcare professionals, and patients alike.
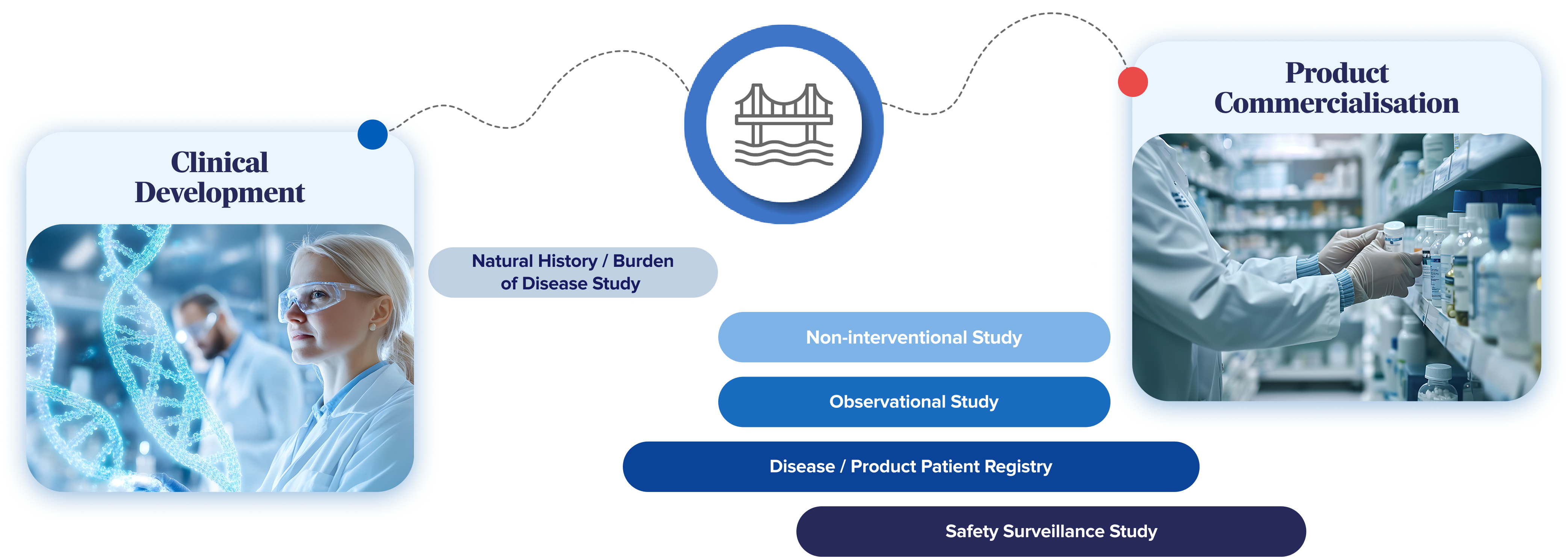

Every project is grounded in our clients' strategic and scientific objectives, with customised solutions to deliver meaningful, high-quality evidence. While each study is unique, our services typically include:
Natural History / Burden of Disease Studies
Mapping the clinical, economic, and humanistic landscape of a disease to provide essential baseline insights and support early product development.
Non-Interventional Studies (NIS)
Delivering high-quality data on safety, effectiveness, and usage under real-world conditions to support regulatory submissions, reimbursement requirements, and label extensions.
Observational Studies
Capturing real-world clinical effectiveness, cost-effectiveness, and patient-reported outcomes through innovative, technology-enabled methodologies designed to reduce participant burden.
Patient Registries
Establishing long-term disease- or product-specific registries to track outcomes, treatment patterns, and sustained impact over time — supporting stakeholders from patients to policymakers.
Safety Surveillance Studies
Generating robust evidence of a product's safety profile in actual practice settings, uncovering insights that may not emerge in controlled trial environments.
Through these services, Optimapharm helps clients demonstrate real-world value, ensuring better decisions, stronger market acceptance, and improved patient outcomes.
Subject Matter Experts

Karol Szczukiewicz
Executive Director, Project Management
A highly experienced clinical research leader with over 17 years in global CROs and pharmaceutical companies, specialising in project and programme management. In his current role, Karol oversees a portfolio of approximately 30 RWE projects across diverse therapeutic areas.

Sylvia Stallinger-Klappert
Project Management Director
With 15+ years in pharma across biotech, pharma, and CROs, she has led 37 RWR studies worldwide since joining Optimapharm in 2020 and co-authored a GI study. Since 2022, she has served as Project Management Director, overseeing PM resourcing and project delivery.

Jeff Trotter
Strategic Advisor
As an entrepreneur, researcher and innovator, Jeff Trotter has been a pioneer in the evolving health economics and outcomes research community and is an industry leader in the design and implementation of patient registries, observational studies, and other RWE (real-world evidence) initiatives.
Resource Hub

I would like to thank you all for the tremendous work done to reach this important milestone for the study in a very challenging and complex global environment. It is the first of many others that we will have to achieve together. Let's keep it up!
Clinical Project Leader, Big Pharma Company
We are very grateful for your hard work and dedication over the past few years. We are delighted to be part of this partnership and look forward to start treating patients in this new therapeutic area soon. We could not have done this without your contribution. We are very excited about the path ahead and can hardly wait to start this study.
Chief Executive Officer, Biotech Company
Very pleased. Looking forward to getting the country up and running soon.
VP Global Clinical Operations, Medical Device Company
On behalf of the entire team, I would like to extend our deepest appreciation and heartfelt thanks for your unwavering support and dedication throughout this phase II clinical study. Your role was critical to this success, and it has been a privilege to work alongside you.
Head of Clinical Operations, Bio-pharma Company
We’re proud to announce the first patient has been enrolled in our Phase 1b/2 trial of a novel, in-house developed drug for colorectal cancer. This marks a key milestone in its clinical development. We thank our dedicated team and our trusted CRO partner, Optimapharm, for their expertise and support.
Chief Medical Officer, Biotech Company

