Turning Science into Hope
Optimapharm is a mid-sized, global Contract Research Organisation turning science into hope through agile, patient-focused clinical development.
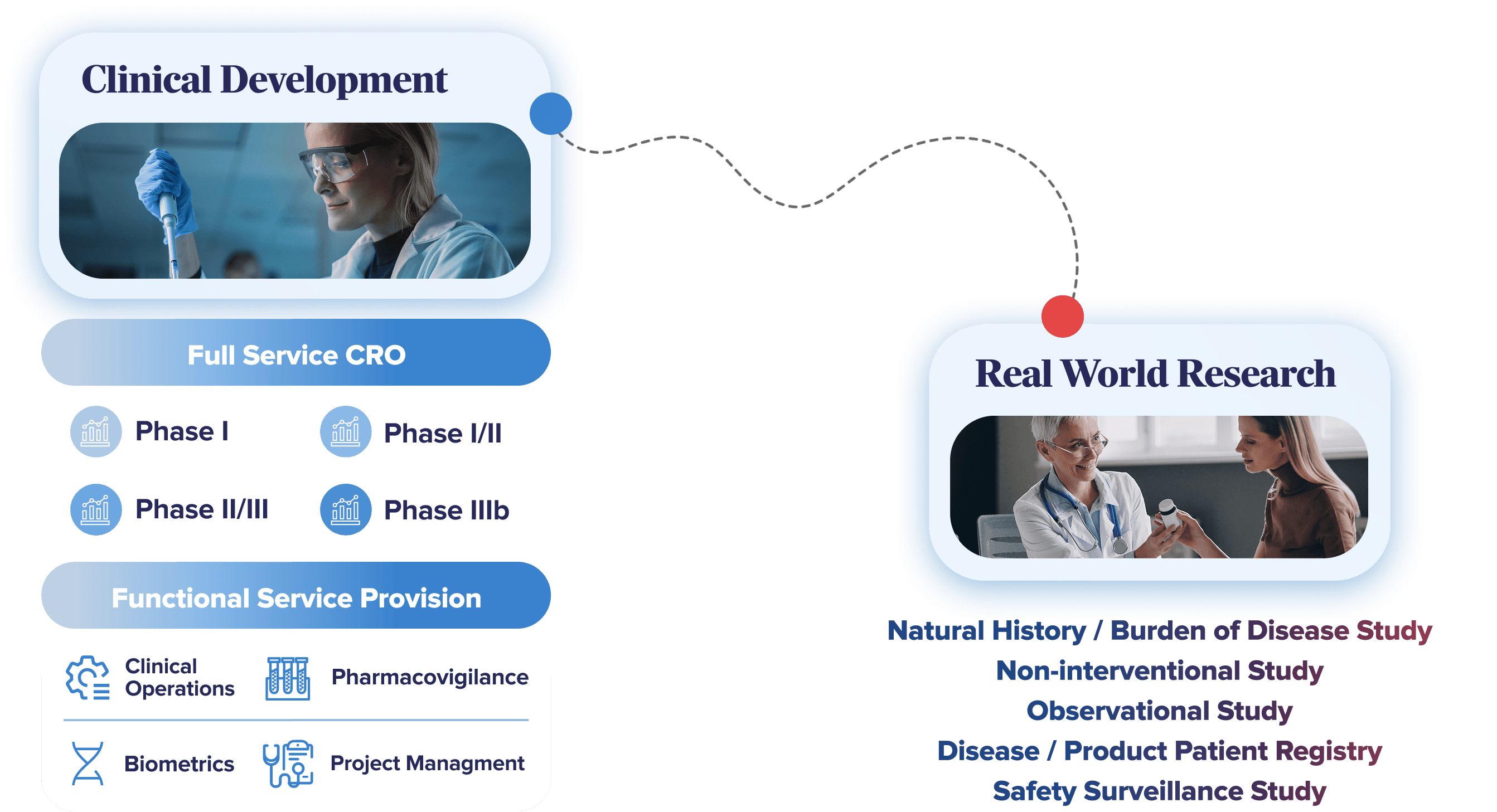
We specialise in Phase I–IV clinical development, delivering Full-Service and Functional Service Provision (FSP) solutions that help sponsors move faster and execute with confidence. Beyond approval, we support continued clinical development and real-world evidence generation to optimise product lifecycles, including repositioning and promotion of approved therapies.

2x more reach across North America and Europe than the average mid-sized CRO
With 2× the patient reach of the average mid-sized CRO and operations in 40+ countries, we enable accelerated regulatory pathways and rapid access to diverse patient populations, helping sponsors shorten study start-up timelines and bring forward first-patient-in.
Built for agility, not bureaucracy, Optimapharm partners closely with sponsors to run smarter, faster, and more efficient trials. Every study we deliver is guided by scientific integrity and a shared purpose to bring meaningful innovation to patients sooner.
"Your role was critical to this trial’s success, and it has been a privilege to work alongside you."— Head of Clinical Operations, Emerging Biotech